Clearing The AirIn Every Operating Room
Cutting Edge Safety Inc.
Breakthrough device to reduce surgical smoke inhalation at site of surgery

Occupational Health Risk
Developed by Professor of Surgery from the University of Chicago, addressing the safety concerns of surgeons inhaling toxic fumes from electro-cautery surgery.
Designed For Surgeons
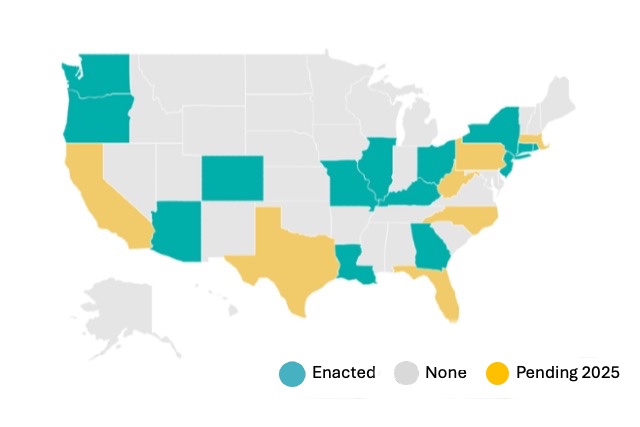
AlleyKat® is designed for surgeons and addresses Occupational Safety and Health Administration (OSHA ) guidelines, currently in force in 20 States.
Patented “Air Curtain"
AlleyKat® creates a unique patented “air curtain” designed to protect patient and surgeon, without obstructing field of view.
Surgical Smoke: A Preventable Risk
What is surgical smoke?
Every time electrocautery or laser devices are used, they generate a visible smoke plume. This plume is more than just an unpleasant odor — it carries ultrafine particles, toxic chemicals, and even viral DNA that can reach deep into the lungs.
What Does The Law Require
Starting in Rhode Island in 2018, legislation has spread quickly: 20 U.S. states now require smoke evacuation systems in hospitals and ambulatory surgery centers (AORN Legislative Tracker, 2025).
Designed By Surgeons
For The Whole OR Team
Headaches, sore throats, coughing, nausea, dizziness, and eye irritation are commonly reported when smoke evacuation is absent (NIOSH, 1996).
What Do The Experts Say
Authorities including NIOSH, OSHA, The Joint Commission, and AORN all agree:
Local smoke evacuation at the point of generation is the most effective protection. PPE is only a backup measure.
Why Does It Matter
A single day of exposure can equal inhaling up to 30 unfiltered cigarettes (Hill, 2012).
DNA from viruses like HPV and HBV has been detected in surgical smoke (Kwak, 2016; Neumann, 2018).
Gynecologic surgeons exposed daily are up to 9× more likely to test positive for HPV DNA in their nasal passages (Wang, 2021).
AlleyKat®: Protecting the Surgical Team
AlleyKat offers a seamless smoke evacuation solution.
Captures plume directly at the source.
Maintains a clear operative field.
Improves visibility safety, and comfort in the OR.
By adopting AlleyKat, hospitals not only meet regulatory requirements but also show a strong commitment to protecting clinicians and patients.
Product
AlleyKat®
Current products do not work well and have not satisfied surgeons for decades
After many years of perseverance, Dr. Millis developed an easy-to-use accessory, using positive and negative pressure air flow across the surgical field to remove harmful surgical fumes.

Our Solution Using AlleyKat®
Smoke is removed directly from the incision site, via a laminar air flow. Surgeons have an uninterrupted field of view, hands can pass through the “air curtain”, fumes are vacuumed away from the surgical field.
.
Simple Plug and Play Set Up
Operating Room (OR) medical air is piped through a sterile tube into the AlleyKat® purpose designed air flow management ring and then attached to standard OR vacuum device which removes smoke. The product is packaged as a kit with all sterile components.
Technical
The graphs depict a smoke simulation experiment. Using an invitro test apparatus, that included a Bovie electrocautery instrument and an Argon Beam Coagulator as a control, raw animal muscle tissue was subjected to a cautery procedure and a particle counter was positioned to measure the particulate count. Data was collected for 2.5 µm and 10.0 µm particle range.
The control apparatus was compared to the test subjects – the results demonstrate a virtual elimination of smoke while using AlleyKat®.
Bovie 2.5 Micron Particles

Bovie 10 Micron Particles

Argon Beam 2.5 Micron Particles

Agron Beam 10 Micron Particles

Regulatory
The product will be filed as a Food and Drug Administration (FDA) Class II, 510k referencing a predicate smoke reduction device, a currently approved device category.
The pathway to approval is estimated to be approximately 12 months.
The product will be packaged as a sterile kit, containing the AlleyKat® air flow management ring, inlet and outlet tubing and universal connectors.

Market
 Since the 1920’s, upon the invention of the Bovie electro-surgical knife, smoke emanating from a surgical field has been a health and safety issue. It is known that the smoke contains a combination of toxic chemicals and carcinogens – all of which can cause long term health complications. Various solutions have been tried but all have failed because of their smoke clearance effectiveness and/or obscuring the surgical site e.g. Green Bovie – which incorporates a smoke evacuation tube at point of use.
Since the 1920’s, upon the invention of the Bovie electro-surgical knife, smoke emanating from a surgical field has been a health and safety issue. It is known that the smoke contains a combination of toxic chemicals and carcinogens – all of which can cause long term health complications. Various solutions have been tried but all have failed because of their smoke clearance effectiveness and/or obscuring the surgical site e.g. Green Bovie – which incorporates a smoke evacuation tube at point of use.
AlleyKat® provides for an uninterrupted view of the surgical site and relies on creating an air curtain that acts a drape capturing all the smoke from the surgical cavity. The price point for AlleyKat® is on par with the Green Bovie, presenting a strong and compelling value proposition.
Over 50 million Bovies are used per year, and with the additional use of argon beam coagulators and electro-cautery ligature devices, the market for open in-surgery cases is estimated to be more than $2.0 billion.
The go-to-market assumption would be to focus on large centers of excellence of thoracic and abdominal surgery, progressively recruiting new sites, leveraging user sites as testimonials and references.
Initial market research not only confirms a large unmet need, the current OSHA Respiratory Protection Standard (29 CHR 1910.134) what has been adopted by 20 states, further supports the Company’s market adoption assumptions.
Company
Dr. Michael Millis, Professor and Vice Chair of Global Surgery at the University of Chicago invented the AlleyKat® device, a unique surgical smoke mitigation device. With the assistance of the University, a USA patent was filed, and following a successful founder’s investor round of funding, Cutting Edge Safety, Inc. was formed.
Board & Management

Phil Croxford, M.B.A.
CEO, Board

J. Michael Millis, M.D.
Chair. President, Founder, Inventor

Terry Winters, Ph.D.
Co-Founder, Board
Venture Capital >30 yrs experience. Ph.D. in chemistry – University of
Wales

Steve Kahn, B.S.
RA/QA

Greg Haas, M.S.
R&D / Operations
>30 yrs experience in Medical Device R&D and Operations

Phil Croxford, M.B.A.
CEO, Board

J. Michael Millis, M.D
Chair. President, Founder, Inventor

Terry Winters, Ph.D.
Co-Founder, Board
Venture Capital >30 yrs experience. Ph.D. in chemistry – University of
Wales

Steve Kahn, B.S
RA/QA

Greg Hass
R&D / Operations
Investors
CES has secured a Seed Round of investment that will take the company to FDA approval within 12 months. Shortly, the Company will be raising additional funds as part of its Series A Round to support post approval commercialization. Interested parties who would wish to participate in this round should contact the Company.
AlleyKat® has a USA patent and applications in other key international markets have been filed.
The company’s technology was developed by Dr Millis at the University of Chicago and the company has secured an exclusive worldwide license for the relevant patents.
